Late last year an article about Pelvic Inflammatory Disease (PID) and associations with endometriosis was discussed online in Nancy’s Nook Endometriosis Discussion and Education Group. The article led me to recall a discussion with my primary care provider a few years ago. That discussion came during an office visit while reviewing intraoperative photographs of extensive thick white adhesions and scarring of the right upper abdomen, liver and diaphragm.
I know I’m not the first woman approached by a medical provider about PID. The topic caught me off guard. I knew about Fitz-Hugh Curtis Syndrome (FHCS) and its relationship to Pelvic Inflammatory Disease (PID) from my graduate studies. I never imagined being questioned about my own intimate behaviors and choices.
Given the professionalism of my provider, and his rapid dismissal of the possibility, I was blessed not to face a barrage of questions with inquisitive tone and facial expressions other women have, and will experience. However, I had brief, internal feelings of embarrassment, uncleanliness and shame even though my personal behaviors and actions have never been conducive to acquire a Sexually Transmitted Disease (STD). I also knew my provider was not passing judgment or perceived me in any way to suggest I was a risk taker with multiple partners. However, about a week after this recalled memory, I experienced a second incident that centered around lack of awareness and misconceptions of endometriosis.
Social media platforms are great methods to amplify information and newsworthy material. Regrettably they create equal opportunity to distribute erroneous information. A lot of users post or respond based upon their perceptions, which are created through their experiences and information they have received (accurate or inaccurate). Through social media, I reached out to a politician in regard to women’s health funding and deficiencies specific to endometriosis. A response from a member of the public stated the disease could be ‘cured’ with ‘moral behavior’ and ‘better dietary choices’. It was also implied, those with endometriosis are 100% responsible for developing the disease. I have no words for what I felt at the moment. But…I can do something.
The recalled memory of my experience with a medical provider and written exchange with a member of the public prompts me to raise awareness about diaphragmatic endometriosis. Specifically, endometriosis of the diaphragm should be a differential diagnosis among females who present with right upper quadrant abdominal pain (with or without referred right shoulder pain) and/or adhesions viewed with imaging or surgery at the diaphragm and liver.
Advocates continue to speak out to correct the myriad persistent myths and misconceptions that stigmatize and cause not only physical harm through delay and appropriate care, but also mental and psychological harm. An average 7-to-10 year delay (some decades) to diagnosis is unacceptable. To add further insult to those with endometriosis, the absurd premise still perpetuated today; persons with endometriosis are 100% to blame for their disease. Uninformed members of society still perceive those with endometriosis ‘had it coming’ and ‘reap what they sow’ through lifestyle choices and experiences. Among the more common myths: amoral sexual behaviors, sexual abuse, poor diet, exercise and stress management. The majority of healthcare organizations, associations and societies continue to disperse outdated, inaccurate information, myths and misinformation that reinforce misconceptions. These are all issues which harm the well being of women with endometriosis.
It’s important to clarify that PID and development of FHCS has occurred in ways other than unprotected intercourse. A few other ways documented include: insertion of an IUD, post-renal transplant with mycoplasma hominis infection, tubal ligation and appendectomy.(1,2,3) Although FHCS is more common among young female menstruators, it has occurred in women post-menopause,(2) and among men.(4,5,6) FHCS-like adhesions can also occur with Systemic Lupus Erythrematosis, Familial Mediterranean Fever (FMF),(1,2,7) Peritonitis, Acute Cholecystitis and Carcinomatosis(5) can present with FHCS-like adhesions. Not all cases with FHCS have been able to determine its original cause. (5,6)
What exactly is FHCS and why should healthcare providers and the endometriosis community know more about it? (continuation from Trish’s Blog #3)
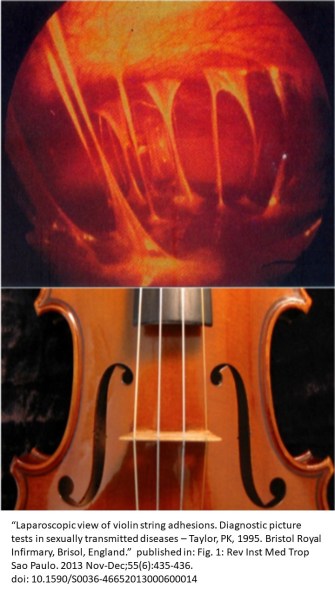
“Fitz-Hugh-Curtis Syndrome (FHCS) is a clinical entity characterized by inflammation of the liver capsule associated with genital tract infection.”(8) Membranes of the abdominal cavity (peritoneum), liver (perihepatitis) and diaphragm may occur as a result of bacterial infection.(9) The bacteria most frequently responsible is Chlamydia trachomatis and less frequent, Neisseria Gonorrhea.(1 ) FHCS consists of an acute and chronic phase. During the acute phase, purulent, fibrinous exudates are present. This can be seen with CT enhanced with Contrast. During the chronic phase there is capsular thickening and adhesion formation with ‘violin strings’ appearance.(1) The Center for Disease Control recently reported that STD’s are on the rise. STD’s lead to PID among females. Among those with PID, a range of 4-27% are estimated to develop FHCS.(9-12)
Symptoms: During the acute phase of FHCS constitutional symptoms may or may not be present. These include: chills, fever, headache, night sweats, malaise. More focal presentations include: nausea, vomiting, vaginal discharge (urethral discharge in a male), right upper quadrant abdominal pain and Tenderness w/wo lower abdominal pain/tenderness. Less frequent are right-sided pleuritic pain, urinary symptoms (frequency or sensation of incomplete void) and hiccups.(8-10)
It is common for constitutional symptoms to be absent. It is also common for lower abdominal pain to precede, proceed or present concurrent with R Upper Quadrant pain and tenderness.
After the acute period, most symptoms are stimulated by tension on adhesions that have developed. These symptoms often coincide with movement, specific postures, coughing, breathing patterns and intensity.(1,11) A similar reduced mobility of the reproductive organs may also be present with internal exam.
Diagnosis: During the acute infectious stage, blood values may be normal or elevated WBC, ESR, CPR, Liver Values, + polymerase chain reaction tests of vaginal secretions for Chlamydia trachomatis and/or N. gonorrhoeae.
Analysis of CT imaging features of 19 confirmed cases of FHCS demonstrate the capacities of enhanced CT imaging to detect abnormalities, dependent up disease stage.(8)
“CT manifestations are closely related to pathological FHCS staging. Acute fibrinous inflammation of the liver capsule surface was seen in the acute phase of FHCS, and MSCT examinations often show no obvious abnormalities. The FHCS pathological changes of the chronic phase were liver capsule fibrosis, hyaline degeneration, localized or widespread liver capsule thickening, and band adhesions between the liver surface and the abdominal wall. MSCT scanning can display homogeneous or inhomogeneous thickening of the liver capsule with or without hepatic subcapsular effusion or pleural effusion, but it cannot display the disease extent or whether local fiber adhesions exist and to what degree.” (8) – Je Sung You et al (2012)
Treatment: FHCS can be completely resolved with Antibiotic Therapy (IV), analgesics and surgical adhesiolysis as needed.
Differential Diagnosis:
| Appendicitis (9,10) | Familial Mediterranean Fever (FMF) (7) | Pleurisy (10,11) |
| Bornholm Disease – (10)
Enteroviral Epidemic Pleurodynia |
Herpes Zoster Infection (8-10) | Pneumonia (Bacterial/Fungal/Viral) (9,10) |
| Cholelithiasis (8-10) | Hepatic Abscess (8) | Pulmonary Embolism (10) |
| Cholecystitis (8-10) | Hepatitis (9,10) | Pyelonephritis (Acute) (8=11) |
| Duodenal/Peptic Ulcer (8,10) | Nephrolithiasis (9,10) | Rib Fracture (10) |
| Endometriosis (1,2,13) | Pancreatitis (9,10) | Subphrenic Abscess (8,10) |
Pathogenesis: FHCS is a secondary condition due to PID from an STD. The mechanism for infection to develop around the liver is unknown. Three theories exist. The first, and oldest theory is through direct dispersal from the reproductive organs, through the peritoneal fluid (1953).(9) However, the lack of infection throughout the abdomen, (specifically)located within the pelvic region and reproductive organs, and most-specific, to the capsule of the liver led to more recent hypothesis of a blood (1992)(9) and/or lymphatic spread (1985)(9) of the infectious agent and potentially aberrant immune response.(6-8)
Note: As an advocate of endometriosis, similarities among the theories of pathogenesis must be clarified. The original theory of pathogenesis for FHCS and endometriosis focused on direct distribution (endo=menstrual reflex theory). Scientific literature is more supportive of blood and lymphatic distribution of pathogen and less focus is given to the theory of direct distribution in development of FHCS. It would be helpful to the endometriosis community to embrace alternate theories of pathogenesis in a similar fashion.
It was not until 2005 a case of diaphragmatic endometriosis was examined for its likeness and potential misdiagnosis as FHCS secondary to PID.(13)
During a data base review of open access articles, only a few articles include endometriosis as a differential diagnoses for FHCS.(1,2) The fact that a large portion of those in the acute stages of FHCS do not present with constitutional symptoms (fever, nausea, vomiting, headache, night sweats, malaise…) and frequently present with clinical attributes among those with endometriosis, (ie. pain/tenderness of the lower abdominal, right upper quadrant; tenderness, decreased mobility of the reproductive organs on pelvic examination, pleuritic pain and referral to the chest, shoulder etc.), this topic deserves greater exploration.
In addition, endometriosis lesions present in various forms (ie. nodules, plaques). The color of lesions are vast and reflective of age and progression of disease. The quantity of glands, stroma, smooth muscle and fibrotic content is wide-ranging.(14) It is important to recognize variability and consider areas which appear as white scar tissue may contain residual active disease beneath the scarring. Excision of these areas are important to remove active disease and restore excursion and efficiency of the diaphragm’s function. A critical feature for differential diagnostics of diaphragmatic endometriosis is a cyclicity and recurrence. Initial presentation of the disease occurs around peri-menses (catamenial). Progress of disease expands recurrent, cyclic symptomatic periods to include both peri-menses and ovulation. For some, symptom become continuous with peaks at peri-menses and ovulation, especially as adhesions and fibrosis advance.
| Signs and Symptoms Differential Diagnosis FHCS vs DE | ||
| Signs/Symptoms | FHCS | Diaphragm
Endometriosis |
| Fever, Headache, Chills, Night Sweats | Y/N | N |
| Malaise | Y/N | Y/N |
| Nausea/Vomiting | Y/N | Y/N |
| Heartburn | N | Y/N |
| Pleurisy | Rare | Common |
| R UQ Pain | Y | Y/N |
| R UQ Tenderness | Y | Y/N |
| Localized Pain R Chest – Pleuritic with movement and respiration. | Y/N Chronic Stage | Common |
| Referred Pain R Shoulder | Sometimes | Common |
| Referred Pain R Neck | N | Common |
| Shortness of Breath | N | Y/N |
| Dyspnea | N | Y/N |
| Catamenial (recurrent, cyclic Peak intensity at menses. It may be present throughout the month. Also occurs in women postmenopause, and without a menses (ie. ocp, hysterectomy) | N | Y* |
| Clinical Tests: Blood, Serum and Imaging FHCS vs DE | ||
| WBC | Often | Sometimes |
| ESR | Often | N |
| CRP | Sometimes | N |
| Bacterial/Fungal | Y | N |
| Enhanced CT | Y
(findings dependent upon disease staging) |
Y/N
(Imaging during symptomatic episode ↑ probability disease is detected. Negative (–) image DOES NOT rule out presence of disease) (MRI > CT sensitivity for diaphragm disease) |
| Dx. of, or symptoms suggestive of abdominal/pelvic endometriosis | Y/N
(Must differentiate sxs. and exam results to menses vs acute PID. Tenderness/ ↓ mobility w/ pelvic exam & palpation of lower abdomen can be present w/ endometriosis & PID) |
Y/N* |
| Hx. of ‘idiopathic’ spontaneous pneumothorax (esp. around menses) | N | Y/N* |
*Yes – Highly suspect for presence of endometriosis lesions of the diaphragm and/or other respiratory tissues.
There are numerous diseases and dysfunctions to consider when a female presents with right upper quadrant pain. Review of FHSC and characteristics of diaphragmatic endometriosis that highlights similarities and differences in their presentations supports inclusion of endometriosis of the diaphragm/liver as a differential diagnosis for right upper quadrant pain.
Citations FHSC vs Diaphragm and Hepatic Endometriosis
Extrapelvic Not Rare Endo Support and Education Group


All Rights Reserved © 2019 Wendy Bingham, DPT Extrapelvic Not Rare
