Where is the liver and what does it do?
- The liver is located on the right side of the upper abdomen and extends a small amount across midline to the left. A large portion of the liver is tucked under the ribcage, just below the diaphragm (which separates the chest and abdominal cavity).
- It is an accessory organ of the digestive system. It produces Bile, a substance necessary for the body to breakdown fatty foods. Bile is then stored in the Gallbladder. It also creates proteins used in other parts of the body.
- The liver is also an excretory organ. The liver and kidney are important to breakdown substances in the body before it is removed. Substances altered by the liver include: some classifications of medications, androgens (sex hormones), alcohol and toxins.
More about the liver’s function’s

Anatomy of the Liver
The liver consists of a right, left and caudate lobe. Each lobe is divided into segments. The larger right lobe is comprised of segments: IV, V, VI, VII,VIII. The smaller left lobe is comprised of segments: II, III. The smallest, caudate, is the third lobe, and segment I. It is located in the posterior liver, overlapping the right and left lobes. The caudate lobe is not pictured in the diagram.
History of Hepatic Endometriosis
Publication History
Hepatic Endometriosis has a very short history. The first recognized published case of hepatic endometriosis is 1986. (1) Over the past three decades, slow accumulation of case studies has created a small collection of information. The exact number of published cases, is unclear. We reviewed 39 cases of hepatic endometriosis in 36 publications. (1-36)
In addition to these 39 cases, another three complete case reports were identified but excluded from the main text of this page. These include two unpublished (37,38) and one published case. The published case of a 70+ yr old person (afab) was excluded as the authors acknowledged immunohistological analysis was not performed to confirm endometriosis.(39) Please note: a few simple characteristics about these three cases are included in a few graphs below. These graphs are located next to but smaller than the graphs which represent the 39 histologically confirmed, published studies (n=39. The graphs are marked (*1) and (n=42). The purpose is primarily to acknowledge a few characteristics that increase the numbers for some characteristics among those with disease. (36-39)
A second set of case reports (6), were identified but excluded for other reasons. These publications are listed at the bottom of the Hepatic Page Citations. Reasons for exclusion include: language barriers, inability to obtain case full text and a single case with a lesion with positive identification of endometriosis components but additional characteristics of the lesion includes other diagnoses . Although excluded, a few important studies and their content deserve brief attention here. The first, Klein F et al’s. 1979 publication “Recurrent spontaneous pneumothorax complicating endometriosis of the liver” predates Finkel et al’s (1986) publication by 7 yr. It is unclear if endometriosis involved the liver surface/capsule or liver body (parenchyma). No known digital copies were located and data bases list locations in Germany for hard copies. Another publication, Sopha SC et al (2015) “Hepatic Uterus-Like Mass Misdiagnosed as Hepatic Abscess” is also excluded. The histopathological report for this case study confirmed the presence of endometrial glands, stroma and smooth muscle. The lesion was described as ‘organoid’, and ‘uterus-like’. To avoid controversy, this case was excluded based upon historical discussions at to whether the lesion could be also be considered an adenomyoma, endomyometriosis, endometriosis or some type of Mullerian tumor (from embryonic tissue intended to develop into organs of the reproductive system).
During our literature review, numerous studies cited 2 publications about the same case. In most of these publications the duplicate case was counted as (2) individual episodes of endometriosis of the liver. The case was published in two different journals with the primary authors (Finkel(1) et al. and Grabb et al.**).
In addition to confusion of recorded incidents of hepatic endometriosis, there are limited prevalence studies, lack of universal methods to collect and record extrapelvic endometriosis (disease present on tissues and organs outside of the reproduction tissues) across all body systems which are barriers to identify true disease prevalence. Recognition is needed that comprehensive data collection and storage of data for locations of all body-wide disease is needed. We emphasize the existence of additional cases, published or unpublished, cannot be dismissed.
Content focus
Hepatic endometriosis may occur in the liver tissue (intra-parenchyma), the perihepatic capsule and surface, or within both areas. The information provided on this page is based upon 39 cases of lesions fully or partially within the liver tissue (parenchyma). Only (1) case of surface endometriosis is included, for a total of (39) cases. These include persons (afab) who are premenstrual (1-5,7,14,16,19-22,25-30,32-35), post menopause. (5,6,8-13,15,17,18,23,24,28) and one unknown menstrual status (36). A persons (afab) with multiple congenital defects of the genitourinary system (10) and one with active disease during pregnancy and post-partum (20) are included.
Reported age at time of diagnosis ranged between between 21 and 62 yrs of age among 39 cases and 21-70+ in 42 cases.(*1) The graph below, lists their age distribution.

The additional (3) subjects yields a slightly different age distribution(*1). (37-40)
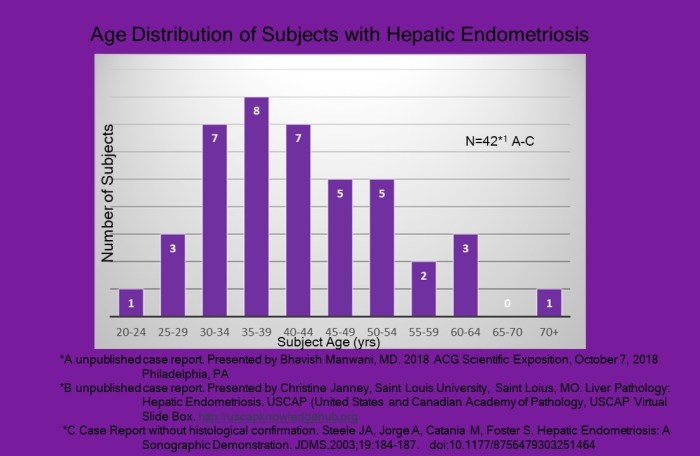
Although many cases included prior diagnosis of endometriosis and pelvic surgery; these, in addition to oral contraceptives use are variables to consider further analysis.
Symptoms of Liver (Hepatic) Endometriosis
Unlike endometriosis lesions that affect other extrapelvic tissues and organs, that occur at the catamenial time frame (around menses). Disease of the liver has been most frequently NOT cyclic. Only a small portion of subjects reported catamenial association (5/38= 13%). (3,25,-27). Only 1 of 2 cases that reported right shoulder pain was cyclical at during the catamenial period.(26) The other case with right shoulder pain reported symptoms as chronic, associated with deep inhalation. (15)
Those with lesions of the superior and/or posterior liver are at risk to development of adhesions between the liver and diaphragm. It is also not uncommon for concurrent lesions of the diaphragm to be present with lesions of the liver or the presence of diaphragm lesions without liver lesions. All three scenarios can create adhesions and present as right upper quadrant/epigastic pain (3,6,15,16,17,21,25,26,34) with or without pain referred to the right shoulder. (15,26)
The qualitative aspects of adhesions that develop between the liver and diaphragm are not acknowledged in the scientific literature. Adhesive disease can be very painful and impacts the diaphragm’s function and a persons quality of life and needs acknowledgement. Adhesive disease can provoke pain with inhalation, coughing, sneezing a variety of body positions and movements. As a contractile skeletal muscle, and primary muscle of respiration, symptoms can appear throughout the menstrual cycle with the act of respiration and movement creating altered mechanics, alignment and increased exertion to contract. (15) The list of impact to Quality of Life is endless.
Lets stop and apply the effects of advancing disease on routine daily events in our lives:
- Positions and transitions: sitting, sidelying, bed mobility, sit-to-stand
- Mobility: walking, stairs; sports participation
- Self Care: dressing, showering, grooming
- Household: meal prep, sweeping, vacuuming, washing windows, driving a car, gardening
- Caring for others (dependents)
- Employment
- Hobbies
All but a single case (38/39) cases involve lesions part or completely within the liver body (parenchyma). Only a single case of the liver surface is included.(19)  Symptomatic complaints include: epigastric/right upper quadrant pain which is acute (1,5,17,23,24,28,30,32,34), cyclic/episodic(2,9,16,18,19,21), chronic(6,8,22,24,33) or catamenial. (3,25-27) A few reported tenderness with palpation (29,33,34); symptoms after meals (18,19), chest pain (28) and malaise. (11,15) Signs of disease (things which can be observed directly or with imaging, lab tests etc.) include: nausea and/or vomiting (1,24,28,30,32); ascites (11,12), jaundice (11,12); portal vein thrombosis (11) and pleural effusion (28). No symptoms were reported in a single case, lesion was found incidently. (31)
Symptomatic complaints include: epigastric/right upper quadrant pain which is acute (1,5,17,23,24,28,30,32,34), cyclic/episodic(2,9,16,18,19,21), chronic(6,8,22,24,33) or catamenial. (3,25-27) A few reported tenderness with palpation (29,33,34); symptoms after meals (18,19), chest pain (28) and malaise. (11,15) Signs of disease (things which can be observed directly or with imaging, lab tests etc.) include: nausea and/or vomiting (1,24,28,30,32); ascites (11,12), jaundice (11,12); portal vein thrombosis (11) and pleural effusion (28). No symptoms were reported in a single case, lesion was found incidently. (31)
Looking for a Diagnosis
Other considerations
A patient’s presentation, their medical and family history is the starting point in the pursuit to find the source of pain and dysfunction. In addition to the liver, a number of other organs can cause  similar symptoms. These organs must be excluded through a variety of clinical tests and an examination. After other organs (ie. gallbladder, stomach, spleen, intestines…) are ruled out, and the liver becomes the primary focus, other conditions specific to the liver must be considered. The results of various tests can help rule in/out the possibility for endometriosis however, tests are not consistently accurate.
similar symptoms. These organs must be excluded through a variety of clinical tests and an examination. After other organs (ie. gallbladder, stomach, spleen, intestines…) are ruled out, and the liver becomes the primary focus, other conditions specific to the liver must be considered. The results of various tests can help rule in/out the possibility for endometriosis however, tests are not consistently accurate.
Can imaging techniques aide
diagnosis of liver endometriosis?
Imaging is used to visualize the structures inside the body. The three most common imaging techniques used to evaluate soft tissue structures, from least to most expensive: Ultrasound (US), Computed Tomography (CT) and Magnetic Resonance Imagery (MRI). Similar to nearly all other areas of the body where endometriosis has presented, no imaging technique is yet sensitive enough to detect all endometriosis lesions, nor specific enough to diagnose endometriosis lesions with an acceptable, low rate of misdiagnosis. The exception is disease of the ovary and structure of the deep pelvic (uterosacral ligaments, between the uterus/cervix/vagina and rectum are trending toward acceptable levels of sensitivity and specificity. (40, 41)
“No clinical or radiological features are specific to this condition, hepatic endometriosis should always be considered on differential diagnosis of women presenting with a hepatic mass, with or without a history of pelvic endometriosis….both pre and post menopause” (36)
– Bhavis Manwani, Mazen Shobassy, Clark Hair (2018)
A complicating factor in the use of imaging as a diagnostic tool for endometriosis is the heterogeneity of lesions.
“Radiologists must be aware that the appearances of endometriotic tissue can vary depending on hormonal response” (7) – Inan M. et al. (2000)
This heterogeneity applies to both structural shape of the lesion (ie plaque, nodule) and the cells which compose the lesions (ie progressive endometriosis lesions composition change as they evolve (glands, stroma, smooth muscle and fibrosis) and in differing quantities (ie. early: glands and stroma late: smooth muscle and fibrosis).
“We looked at the previous reported cases and concluded that there is no magnetic resonance imaging, computer tomography or ultrasound characteristics exclusive, specific to hepatic endometriosis, but the most common imaging feature of hepatic endometriosis in these reported cases are well defined, lobulated, cystic lesions with solid components and septations” (18) – M Asran, A Rashid, J Szklaruk (2010)
The imaging techniques and findings of (39) cases of intraparenchymal hepatic endometriosis can be read here:
Imaging Table Hepatic Cases N=39
Auxilliary Testing
Nearly all cases underwent additional clinical testing to establish a diagnosis. Very few abnormalities were found among tests of Blood Values, Liver Enzymes, Tumour Markers and Antigens/Antibodies for viral and infectious diseases. Findings helped rule out many diagnoses. The use of Angiography and Image-Guided Needle Biopsy provided supportive information about lesion(s).
To view list of auxiliary tests and abnormal findings:
Limitations of imaging to accurately differentiate benign lesions of endometriosis from malignancies, and the low, but existent possible malignant transformation of endometriosis exists. Therefore, surgery remains the primary intervention.
Treatment
The treatments offered for management of endometriosis are limited to medical management, to include hormone suppression and surgery. To date, women continue to be offered surgical castration as a way to ‘manage disease’. It is important to note this technique is no longer recommended for management and prevention of further disease. Surgical care for endometriosis consists of ablation and excision. Ablation is the traditional approach, used since the 1940’s. Ablation can only penetrate 1-2 mm deep, leaving deeper disease untouched. Excision, an older technique that has rebounded in effectiveness due to advanced ability to visualize the operative field, surgical instrumentation, coupled with expanded knowledge about disease characteristics and behaviors. Excision removes the entire lesion. Ablation has been the standard approach to treat endometriosis of the pelvic organs for decades. Unfortunately this approach has poor efficacy. But, lesions in extrapelvic locations have historically been managed differently by most providers. Extrapelvic lesions have most commonly been excised. Why the difference?
It is common for lesions to be misdiagnosed and imaging technology has limited ability to differentiate between benign endometriosis and malignancies. In addition, the composition of endometriosis lesions differ throughout its lifespan. This impacts the appearance on imaging with a broadened range of characteristics. As you will read below, the efficacy of medical management has been very poor among hepatic and other extrapelvic locations.
Medication (Hormone Manipulation)
Literature review found six (6) cases with utilization of hormone suppressants to manage disease. Three were pre menopause (3,5,7,13) and two were post menopause (10,15) (surgically induced 5yrs and 8yrs prior). The effectiveness of hormone suppression on lesions of the liver is not conclusive among the six (6) cases. One publication did not provide follow up after diagnosis and initiation of Danazol. (7) Application of hormone suppressant in the other five (5) cases differ in duration and frequency of treatments. They report no reduction in lesion size (4,10) or reduction but not resolution of lesion (3,15) while on the suppressant and regrowth when discontinued. (13) One case reports resolution of intrahepatic cyst following use of intranasal Goserelin. (10) The results must be viewed cautiously for possible misdiagnosis and concurrent treatment resolved lesion. (10)
A summary of each case with trial of hormone suppressant agents can be accessed here: CasesHormoneSuppressantUseHepaticEndo
Further discussion about the use of GnRH-a among persons (afab) with hepatic endometriosis, among pre menopause and those with surgical castration:
GnRH among Women with Hepatic Endometriosis
Surgical
The majority of cases underwent liver resection. The type of resection was dependent upon location and lesion size. With lesion removal most techniques also removed a small circumference of healthy looking liver tissue around the lesion (clean margins). This is to ensure that the entire endometriosis lesion is removed to prevent regrowth. The degree of resection has included: cystectomy (16) (lesion enclosed by a thick cystic wall preventing endometriosis cells from spreading liver tissue), segmentectomy(ies) (42,9,10,20,30) and lobectomy (hemi hepatectomy) (5,6,9,10,11,12,13,24,29) for lesions which occupy the majority of a liver’s lobe. A single case described Ultrasonic Peri cystectomy. (22) An early case was treated with the lesion ‘deroofed and drained’. A second surgery with cystectomy due to regrowth. (1)
Hepatic Endo in
Pre & Post Menopause Women
Hepatic endometriosis is confirmed in both pre and post menopause persons. 38/39 cases confirmed menstrual status. One case did not disclose the information. This case is excluded. (36)

Premenopause
Among the (24) persons who were pre menopause: seven had a previous diagnosis of endometriosis (4,7,16,25,26,30,34); six denied a history of disease and were asymptomatic (19,20,27,28,29); three were asymptomatic but had confirmed pelvic endometriosis at time of liver resection (2,16,32); four had confirmed absence of pelvic disease from prior laparoscopy or at time of liver resection. (5,21,22,33) No disclosure about presence/absence of pelvic disease in four cases. (1,3,14,35)

Post Menopause
In alignment with endometriosis found in other extrapelvic locations, hepatic endometriosis occurs in persons after menopause. Active disease was identified in fourteen (14) of the (38) cases investigated. (5,6,8-13,15,17,18,23,24,28). One case was reported in a persons (afab) over age 70 yrs. Detailed description of the lesion was included in the case report however, the authors acknowledged failure to get histological confirmation. This case is not included in the graph below (38).
Among the (14) postmenopausal women with active hepatic endometriosis, (11) were surgically induced. (6,8,9,10,11,12,13,15,17,23,28)

Of the (11) persons with surgically induced menopause, ten people had a medical history of confirmed pelvic endometriosis.(6,8,9,10,11,12,13,15,17,18). Four people had no history of endometriosis. (5,23,24,28) Those with surgically induced menopause (n=11), nine had history of endometriosis, two did not.

In regards to Hormone Replacement Therapy (HRT), two reported use at time of diagnosis (8,17), four had remote use (11,13,15,28) and three cases did not use HRT (5,12,24). Use of HRT couldn’t be determined in six cases. (6,9,10,18,23).

“In our laboratory, estradiol level of 22pp/mL is considered ‘castrate levels’. Sources of estrogen in the post-menopausal patient arise from the cytochrome p450 enzyme aromatase. Aromatase is primarily expressed in ovarian granulosa cells, placental syntiotrophoblast, adipose tissue, skin fibroblasts and the brain…. The aromatase system may be one source of estrogen in cases of postmenopausal endometriosis. The endometriotic implants may be producting enough estrogen to maintain endometrial-like activity”. (15)
– JE Jelovsek, C Winans, J Brainard, T Falcone (2004)
Lesion distribution:

Liver volume
distribution and location of hepatic lesions
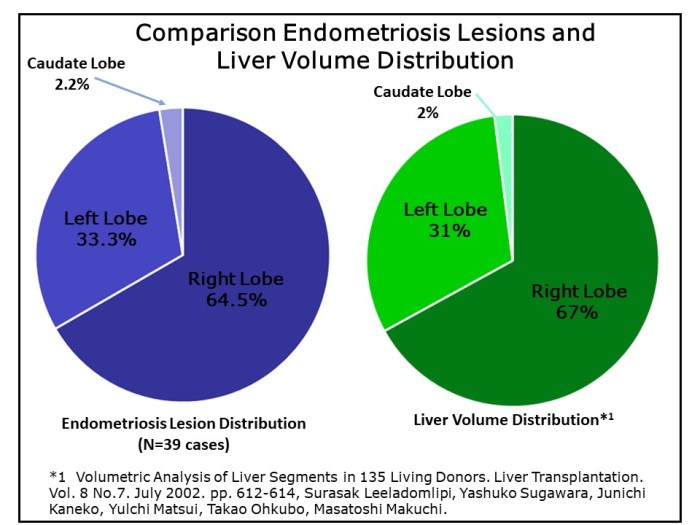
As the number of published cases accumulated with time, there is a shift in locations and, with greater numbers a more likely representation of the disease. As noted above, based upon 39 cases there is a preponderance for greater number involving the right lobe.


If we consider the volume distribution of the liver, the right lobe comprises an estimate 67% of the liver’s total volume. The left and caudate lobe compose the remaining 31% and 2% respectively. (46) When these ratios are compared to the distribution of endometriosis lesion between the liver’s lobes. The distribution among (39 cases) is very similar to volume distribution of the liver. Simply, a homogeneous distribution of lesions with the liver lobes. However, collection of, and analysis of location specific to the segment(s) and each lesions direction (anterior, posterior, superior, inferior) would provide more detailed distribution mapping. Mapping may provide evidence for pathogenesis.
Pathogenesis
Not all authors expressed their hypothesis of disease origin and support or refute any or all of the established theories for disease origin. Some limit their discussion to an overview of each theory and its application to the liver.
“The coexistence of pelvic endometriosis in our patient could suggest dissemination through the clockwise intraabdominal circulation of peritoneal fluid. On the other hand, endometrial cells are not normally present in the physiologic retrograde menstruation…In the embryo the paramesonephric ducts derive from an investigation of coelomic epithelium. Furthermore, this epithelium covers the hepatic ‘bud. Hepatic endometriosis could originate from a spread of mullerian cells in the cranial direction or possibly from coelomic metaplasia of the peritoneum covering the liver.” (2) – Rovati V. et al. (1990)
A few authors have extended their deductions to include support of a particular theory of origin based upon the characteristics of their case. A list of citations which support each theory and a few noteworthy quotes by the writers are provided which support or refute the four fundamental theories: Hematologic and Vascular Dissemination; Metaplasia, Mullerianosis and Retrograde Menstruation.
Hematologic and lympthatic dissemination (3,4,9-11,16,18,27,31)
“In our case, the hepatic cyst was not completely intra-parenchymal and there was no evidence of associated genital lesions….We speculate that the lesion originated from metaplasia of the peritoneum covering the liver or from endometrial fragments transport into the liver by lymphatic or blood vessels”. (3) – Cravello (1996)
“In our case the lesion most probably resulted from endometrial fragments transported into the liver by lymphatic or blood vessels during surgery for pelvic endometriosis eight years previously. Although an origin from the perineum covering the liver cannot be ruled out completely, this possibility is less likely because the cyst was completely intraparenchymal.” (9) – Huang WT (2002)
To understand a little bit more about how things can go wrong during early embryo development and along the lifespan, please read here:
What happens during early embryo development that could possibly go wrong
Metaplasia (3,8)
“Usually, the pathogenesis of endometriosis is explained by the implantation theory or the coelomic metaplasia theory. The former theory postulates the deposition and growth in ectopic localisation of viable endometrial tissue by retrograde menstruation, lymphatic or bloodborn dissemination or iatrogenic transplantation during surgery. The coelomic metaplasia theory states that endometriosis develops from the secondary mullerian system, i.e. the cells lining the pelvic peritoneum which have the potency of metaplastic transformation into endometrial, tubal and cervical epithelium……..
………”Associated pelvic endometriosis was not apparent in our patients, so reflux of endometrial tissue by retrograde menstruation as a pathological possibility of the pancreatic and hepatic endometriosis seems to be improbable. Moreover, as the history of abdominal surgery in the second case dated back from early childhood and as rigorous inquiry revealed that the blunt abdominal trauma in the first case had been a bagatelle injury, the implantation theory can explain
the manifestation of endometrial cysts in liver and pancreas only by vascular dissemination along lymphatics and blood vessels. The metaplastia theory seems to be a more attractive pathogenetic principle in our cases when we assume that the secondary mullerian system comprises the upper abdominal peritoneum as well. This would mean that the peritoneum covering the surface of the liver and lying over the pancreas accomplishes its metaplastic potencies and subsequently proliferates into the organ’s parenchyma (e.g. by tying off the peritoneum on the anterior surface of the pancreas and growing posteriorly into the gland). This mechanism was also mentioned by Marchevsky et al.” (5)– Verbeke C , Harle M, Sturm J (1996)
“The coelomic metaplasia theory states that endometriosis develops from the secondary mullerian system, from the cells lining the pelvic peritoneum which have the potency for metaplastic transformation in endometrial, tubal or cervical lesions. In the present case the hepatic endometriosis was incompletely intraparenchymal and appeared to extend from the periphery to the liver” (8) – N’Senda P. et al. (1999)
The theory which has received the most funding and focus continues to be Retrograde Menstruation. However, all disease cannot be explained by a single theory. Each theory has limitations and imperfections to explain all disease.
In regards to endometriosis identified in organs and tissues remote of the female reproductive system, two other theories are discussed: Vascular/Lymphatic Dissemination and Coelomic Metaplasia. For disease within the pelvis, in close proximity to the female reproductive organs or among most of case reports in those assigned male sex at birth, Mullerianosis is often the focus. However, disease arising in certain remote areas that originate from the same primitive germ layers (mesoderm), Mullerianosis should be considered.
Mullerianosis (15)
Novak and Woodruff (r) reviewed 35 malignancy cases involving extrapelvic endometriosis. All 35 cases were adenocarcinomas. Endometrial stromal sarcomas as well as malignant-mixed mullerian tumors have been reported in the setting of endometriosis but are exceedingly rare. In summary, endometriosis should be considered in the differential of a symptomatic liver mass in premenopausal as well as postmenopausal people. Surgical castration was not curative or preventative in this patient. Malignant transformation of endometriosis is an uncommon but possible event and should prompt serious consideration for resection of detected lesions. This case should stimulate new hypotheses regarding the pathogenesis of this disease.” (15) – Jelovsek JE et.al. (2004)
Retrograde Menstruation
There was no support of Retrograde Menstruation by the authors of any of the (39) cases. Their unanimous dismissal of Sampson’s Theory of Retrograde Menstruation (Implantation Theory) is based upon lesion location within the liver body (intraparenchymal). However many authors were supportive of vascular and lymphatic distribution of disease also proposed by Sampson. (see ‘Hematologic and Lymphatic Distribution’ listed above).
Summary
Documentation of endometriosis involving the liver has only been reported for about 4 decades. During this period, the number of cases has accumulated, and with greater frequency in recent years.
Primary focus of this page concerns endometriosis lesions entirely or partially within the liver tissue (parenchyma). We reviewed 39 cases, of which 38 cases were intra parenchyma. It is important to acknowledge another (3) cases could not be included in statistics and (5) publications could not be reviewed due to language barriers or inability to access. These are (8) potential cases that could raise the total to (47) cases. The existence of undiscovered published and unpublished during our database searches and review of citation lists among all publications accessed.
Endometriosis of the perihepatic capsule/liver surface was not investigated in detail for this page. Numerous published and published cases exist, and likelihood that surface lesions are more common than intraparenchymal lesions. However, disease of the liver’s surface is most often a secondary finding and report among literature with lesions of the diaphragm and pleural or catamenial pneumothorax and hemothorax as the primary focus in publications.
The profile of those with endometriosis of the liver is the same as those with endometriosis elsewhere in the body. The disease does not discriminate in age, ethnicity and whether pre or post menopause. The disease has demonstrated activity during pregnancy and immediate post-partum. (20) The disease is found among those with normal anatomical menstruation and those with both congenital deformities and agenesis of body organs. (10)
Lesions within the liver can range between very small to very large before symptom presentation. Like endometriosis elsewhere, it can also be asymptomatic and similarly, demonstrate variability in response to medical management. It was common for lesion regrowth after discontinued hormone suppression. (4,10) Other findings included no reduction or incomplete resolution of the lesion. (3,15)
Advanced endometriosis of the liver, can have detrimental impact to the individuals health and function of other organs. Like other regions of the body, neighboring organs can be affected. As we have seen, disease of the liver has extended to the bile ducts (5,20), gall bladder (6), diaphragm (3,6,8,13,15,16,17,20,21,24,25,26,34), pericardium (17), heart (21), lung parenchyma (20),and retroperitoneal space. (10,12) Not included in this page, the impact of disease of the perihepatic capsule and liver surface also contribute to organ dysfunction which must not be excluded.
Conclusion and future considerations
- The pathogenesis of endometriosis remains unclear.
- Possibility more than a single origin to disease development exists.
- Detailed documentation of segment and directional location of each lesion may help further identify evolution of the disease.
- Investigation specific to pericapsular/surface lesions of the liver deserve analysis in similar format.
“Hepatic endometrioma should be included in the differential diagnoses for a woman of any age presenting with a hepatic mass with or without a history of pelvic endometriosis”. – Goldsmith (2009) (17)
Hepatic Citations
(Total potential cases known to-date for intrahepatic endometriosis: (47) (August, 2019)
Extrapelvic Not Rare Education and Discussion Group


All rights reserved ©2019 Wendy Bingham, DPT. Extrapelvic Not Rare
