No one really knows how many women suffer with endometriosis, but a good estimate is 176 million. Even less is known about the number of men who have undiagnosed and unreported endometriosis. Since Oliker and Harris published “Endometriosis of the bladder in a male patient” in the Journal of Urology in 1971, there are about 20 case studies in the scientific community. The majority of these cases have two things in common:
- The endometriosis lesion occur along the path of cells laid down during early embryo and fetus development. The majority of these cells normally ‘regress’ (disappear) before being born. However, it’s normal for a small portion of cells to remain after birth. These cells are inactive, dormant. They are referred to as embryonic cell rests. In development of endometriosis lesions among males, it’s suggested these cells are stimulated and continue to differentiate. Lesions that develop into ‘endometriosis’ present with specific characteristics to include glands, stroma and hormone receptivity to estrogen and progesterone. (1)
- Elevated circulating estrogen is suggested as a stimulating factor in most cases. Among these cases, elevated estrogen occurs through one or more of these three mechanisms:
a.) Exposure to estrogen compounds for treatment of prostate cancer (2-7)
b.) Cirrhosis of the liver (8-9)
c.) High body mass index (obesity).(10-11)
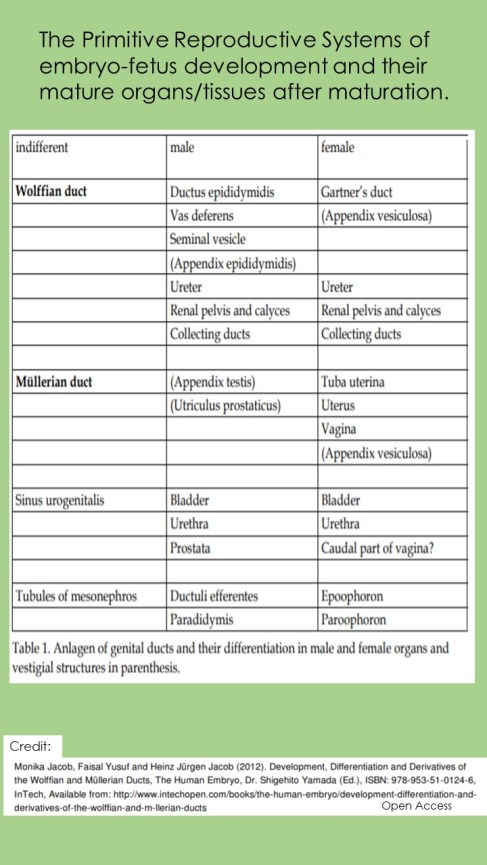
1.) To expand on item #1 above: Before 8 wks of fetus development, tracks of cells migrate to a lower position in the fetus and replicate as the primitive urinary and genital system. The first track is the Wolffian Ducts (WD). This track primarily creates the male reproductive system. A second track of cells Mullerian Ducts (MD) develop shortly behind the first track. It primarily creates the female reproductive system. As the embryo develops into a fetus, presence or absence of the ‘Y’ chromosome impacts which of these two tracts continues to develop and which regresses (XY=Male, XX=Female). Hence the WDs are influential in the fate of the MD. (1)
If the ‘Y’ chromosome is present, cells (Sertoli cells) from this first track (WD) produce a substance with a single purpose (AMH). Sertoli cells secrete Anti-Mullerian Hormone (AMH). Cells in the MD are sensitive to AMH. Response to AMH normally leads to death or regression of these cells and development ceases among Mullerian cells programmed to become female reproductive tissues and organs (uterus, fallopian tubes and vagina). It’s normal, among both sexes, to have some embryonic cell rests after birth and throughout our lifespan. These embryonic cell rests normally remain dormant (inactive). (1)

Things get complicated from here. There is a specific ‘window of opportunity’ when the cells of the second track (Mullerian – primarily female reproductive system) are sensitive to AMH. This window is open from about the 8th week to the 4th month of fetus development. (1)
Among males, there are two Mullerian (MD) remnants which are normally present in adults: appendices testis and prostatic utricles. (1) However, if there is an incongruency between the rate of development in the second track (MD) and release of this substance (AMH) by the first tract (WD), it can cause primitive cells to further develop into female tissues and organs of reproduction. At the far end of development, they may become immature ‘uterus’, gonad(s) (ovaries) etc., in a male who may have normal, functional male external genitalia. Aberrant timing of AMH released outside the sensitive time period, instead of promoting cells death, the cells remain and/or continue to develop (Persistent Mullerian Duct Syndrome – PMDS). (1) Causes for altered release of AMH is exposure of the mother to environmental toxins and endocrine disruptors. (15) 
Locations of endometriosis in males has been reported at the bladder (3,6-7,10), inguinal canal adjacent to the spermatic cord (Mf, 8-9,12), epididymis (14), vas deferens (11) testes (2,13,16), prostate and abdominal wall. (5)
(Alternate theory) The authors of a few case reports suggest an alternate mechanism for the growth of endometriosis lesions based upon the location (5) and/or histological aspects of their cases. (16) In these cases it is proposed that endometriosis lesions developed through transition of cells from one type of ‘specialized cell’ to another (metaplasia) among older observations. (16) Today, the conversion of mature ‘specialized’ cells would be called ‘transdifferentiation’. Metaplasia is currently considered to be replacement of one ‘specialized cell’ by another through stem cell differentiation.
“In our case, we didn’t find any Mullerian duct remnants such as appendix testes and paradidymis which could support embryonic rest theory.”
“The endometrial type epithelium showed continuity with a mesothelial cell layer. The continuity strongly supports a metaplastic genesis of the endometriosis in the present case.”
“Although the tunica vaginalis is among locations for Mullerian duct remnants to become stimulated, this paretesticular lesion presented histochemical composition suggestive of metaplasia of mesothelium to endometriod epithelium.”(16) – Zamecnik M et al. (2013)
What are specialized cells? All cells are created from stem cells. Stem cells compose the initial three primitive germ layers formed during early embryo development, and from which all organs and systems develop . These primitive germ layers are the endoderm, mesoderm and ectoderm. For this discussion, focus is on the mesoderm. It is from the mesoderm that the skeletal bones, connective tissues, muscles, urogenital systems and lining other internal body cavities develop. These are end specialized cells.
During embryogenesis (fertilization to 8th week) cells become progressively more specialized. The composition of cells become task specific to fit its specific role within a tissue or organ (ie. A person wears specific clothing and tools dependent upon the job: a chef wears apron, hat and uses spatula, knives, bowls, kitchen appliances etc. A Construction worker wears steel-toed boots, knee pads, gloves and handles shovels, works heavy equipment).
Among cases without predisposing factors (Mullerian remnants, history of female hormone use, cirrhosis or obesity), endometriosis among males centers around another theory (coelomic metaplasia) to explain lesions which appear to develop from one type of specialized cells and convert to another specialized cell (endometriosis lesion). The common factor is that both specialized cells are from the same type of stem cell – mesoderm. (15)
Sampson’s theory of retrograde menstruation and lympthatic/vascular spread with uterus as location of origin is excluded due to a lack of mature functional female uteri.
2.) To expand on item #2: Although the number of endometriosis case reports in human males is very limited (abt. 20), most reported cases of male endometriosis suggest that stimulation of embryonic rests through elevated circulating levels of estrogen trigger development of endometriosis lesions from embryonic rest cells or more developed Mullerian Duct Remnants. These include: a.) Hormone treatment following prostate cancer b.) liver cirrhosis and c.) high body mass index (obesity).
a.) Hormone Treatment (2-7):
After surgical removal of malignant primary tumors of the prostate, those with metastatic tumors are often treated with long duration female hormones to shrink these tumors. Although a small amount of estrogen is normal within human males, their use further elevates their levels in circulation. It is suggested greater levels stimulate residual embryonic rests (2-3,5-7) or more advanced Mullerian Duct Remnants to grow.
b.) Liver Cirrhosis (8-9,17):
“Cirrhosis of the liver in men creates altered production of estrone (E1) and estradiol (E2) to levels found in women.”
“As clearances rates for androsterone remains unchanged but conversion from testosterone increases causes increased blood level of androsterone , the clearannce rate of testosterone decreases but plasma levels dont increase because testosterone is converted faster to androsterone (E1) so testosterone levels stay normal”. (17) – Gordon GG et al. (1975)
Two cases of males with history of liver cirrhosis, each underwent multiple surgeries to resolve a Right inguinal hernia. Although it is recognized the inguinal area lies on the path of both Mullerian and Wolffian Duct Systems, where dormant embryonic cell rests may be present, One research team suggested endometriosis lesion developed through metaplasia of local area tissue prompted by elevated estrogen levels.(8) In contrast, the other research team conclude that the repetitive post-operative inflammatory environment and elevated estrogen levels stimulated embryonic rest cells. (9) (Note: Neither study reported confirmation of residual embryonic cell rests or histopathology to support/refute their conclusions).
c.) High body Mass Index (Obesity) (10-11,16):
Elevated levels of estrone (E1) and estradiol (E2) through testosterone conversion (aromatization) appears to be …”increased in obese men in proportion to the percentage above ideal weight.” (18) – Schneider G. et al. (1979)
The researchers in the case of an 46 yr old, obese male with surgically confirmed removal of endometriosis cells from the tunica vaginalis (along the Mullerian Duct pathway) suggest the lesion was not the result of stimulated embryonic rests, as no Mullerian Ducts were present. Based upon tissue samples with confirmed estrogen and progesterone receptors and mesothelial cell aspects (calretinin and cytokeratin 5/6), transformation from one specialized cell to another occurred (metaplasia) which may have been promoted through elevated estrogen levels known with obesity. (16)
To contrast…
“In relation to male endometriosis it could be theorized that (this) aromatization could provide pathological elevated estrogen levels to drive growth of endometriosis from remnant embryological cells in a male”. (11) – Rei C. et al (2018)
Researchers of a 54 yo obese male with gross hematuria (blood in the urine) report endometriosis of the bladder wall. The male did not have a history of cirrhosis or cancer with exposure to female hormones. It is acknowledged that the Mullerian Duct System involves the lateral aspect of the bladder. Although the publication does not report observation of mullerian ducts (excision of lesion was transurethral – from within the bladder, thus no view inside pelvic cavity) the authors suggest activation of embryonic rest cells.(10)
What about the minority of cases?
Some cases cannot be fully explained. One of these include a 21 year old male with normal male reproductive organ maturity. (19) He spontaneously developed lower abdominal pain. Imaging and surgical specimens determined ‘pseudohermaphroidism‘. An immature uterus, and gonads were present. It is understood that the uterus, fallopian tubes and vagina are Mullerian cells (which are formed under direction of the Wolffian System) however the gonads (ovaries of females and testes of males) develop independent of the Wolffian system. (1) The present case reported male Vas Deferences attached to each internal gonad, without clarification of any advanced development of these gonad and hormone synthesis. There was not reports of estrogen blood levels. The three primary estrogen elevating conditions were not applicable (liver cirrhosis, obesity or prostate cancer).
Another case. A healthy 27 yo. male with unremarkable medical history complained of scrotal pain for two weeks.(14) After ultrasound confirmation, a small mass of the left epididymis was removed with local anesthesia. The mass was confirmed as endometriosis. A genetic screen found normal 46 chromosome XY, normal hormone levels and endocrine function. There was no obesity, cirrhosis or prior use of steroids. The research team commented that:
“About 1% of males have persistent Mullerian Duct remnants (persistent mullerian duct syndrome-PMDS). Normally in the second month after fertilization, a substance is released that stops production of growth into the opposite sex organs. From days 55-76 the cells of Mullerian Ducts become less sensitive to this substance. As a result, a continuous development into genitalia may occur. Normally this only occurs with 46, XY Syndrome in which the person continues development as a male normal structures but with internal infantile uterus and fallopian tubes. Secondary mutation in MIS gene (Mullerian Inhibitory Substance) hence no substance is ever produced to allow male genital growth to continue while inhibiting further development of female organs. (14) – Giannarini G et al. (2006)
A lack of collective circumstances in this case does not provide support of any particular theory of origin in this case:
“The development of an endometriosis cyst with morphological features of hormonal stimulation is an event that remains difficult to explain, given that our young patient had normal endocrine function and denied use of steroids”. (14) – Giannarini G. et al. (2006)
Another two men (aged 43 and 50 yrs old) with 2cm testicular masses (ie obese, cirrhosis etc.?) which were confirmed endometriosis upon surgical excision. The lesions occurred along the primitive reproductive system ducts, likely developed from embryonic rests. It may simply be omission of information to clarify the health status of each man in their case studies, but there are no explanations to what or how these lesions were stimulated to develop. (13)
The last and most recent case involves a 66 year old male diagnosed with a 3mm endometriosis lesion of the interior surface lining of the bladder. (21) He, too, did not fit the established profile of people amab for risk of endometriosis. However his medical history included two autoimmune conditions (psoriasis and ulcerative colitis) and recently completed a (03) weekly intravenous rounds of chemotherapy shortly after surgical resection of all non-invasive malignant bladder tumours. The endometriosis lesion was identified near the entrance of the left ureter. The authors of this case present arguments for:
a.) metaplasia of naturally occurring cells that have transitioned from one type of cell into another as a result of hormonal and inflammatory changes and…
b.) possibility the endometriosis lesion arose from ’embryonic rests that normally regress and die off during early embryo development. (primitive reproductive tracts of both identified sexes (male and female) begin development during early development of the embryo. If the embryo contains a Y chromosome, a chain of events leads to cessation (in further development of the reproductive organs associated with persons afab. These tissues either ‘die off’ or become ‘dormant’ as Muellerian Remnants’).
Given the limited application of the chemotherapy agent in this case, despite its oestrogenic enhancing and anti-androgenic properties coupled with the individuals normal hormone blood levels, the authors acknowledge it as a factor in the growth of the endometriosis (it does not explain the development of the lesion in the person amab. Sarkar et al remark “Whether this immunological factor is an isolated cause or a synergistic factor to one or more of the other causes is yet to be determined”. (21) To paraphrase, its unclear if the altered ability of the immune system (both systemic and local immune integrity) are a causative factor in the INITIAL establishment of lesions OR its deficiencies allow lesions to grow.
Review of factors, urinary and reproductive system development and case reports, does it appear that endometriosis among males is a different disease than that among women?
It is strongly suggested that the disease has multiple origins and types of disease. There is certainty the disease is identified among human female fetuses and numerous examples of lesions have been identified as displaced endometrial glands and embryonic ducts which demonstrates one probable origin of disease in which disease evolves from in both females and males. (20)
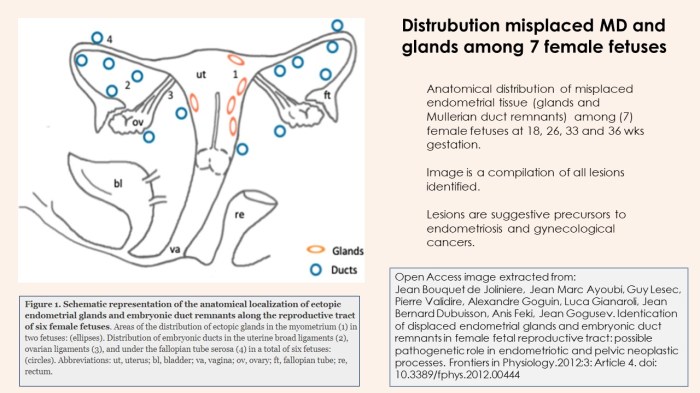
At first glance, if you did not realize this was an image of lesions among fetuses, one may assume its an image of retrograde menstruation among a menstruator. The majority of practitioners tend to follow Sampson’s Theory of Retrograde Menstruation. Perhaps its time to invest more funding into the alternative theories. Certainly there is objective evidence to support this movement.
Citations: Endometriosis in People AMAB
Last Update: 08/30/2024
All Rights Reserved © 2019 Wendy Bingham, DPT Extrapelvic Not Rare
