Terms to Know
Catamenial: defines a time period around the start of the monthly menses. The older, and larger period extends 72 hours prior to, and 72 hours after the start of menses. The newer, smaller period extends 24 hours prior to, and 72 hrs after menses starts.
Manifestation: is a physical process that can be measured. It signifies the presence of a pathology or trauma. Manifestations of thoracic endometriosis are referred to as ‘Thoracic Endometriosis Syndrome’ (TES).1
Thoracic Endometriosis Syndrome (TES): includes physical changes that can be quantified (seen directly by a clinician,through imaging or syringe extraction). The four traditional manifestations, and estimate prevalences include:

Although not specified, a portion of persons with spontaneous pneumothorax (SPT) due to endometriosis, occur outside the ‘catamenial’ period (non-catamenial/endometriosis related pneumothorax, ERP). ERP is included in TES. 2-4
The addition of Catamenial Pleural Effusion, Diaphragm Herniation and Catamenial Chest Pain (CCP) have been suggested as manifestations of TES.5 Although Catamenial Chest Pain (CPP) is not a manifestation, a large portion of those with TE experience it, which can become incapacitating.

With exception to preventive measures, invasive procedures are not performed without direct evidence or accumulation of findings that reach a level of probability that suggests disease is present. Three categories of evidence contribute to establishing a diagnosis: Objective; Observation by the Provider; Subjective-Objective provided by the patient.
Objective: Blood and urine Tests, imaging, histological samples and cultures, endoscopic observations, body temperature, reflexes, neurological exam components, cognitive testing.
Observation: information gathered by the clinician may include: color of skin, presence of perspiration, disposition of patient, mobility and interpersonal communication.
Subjective-Objective: information gathered from the patient (past medical history, family history, history with current symptoms that include their behaviors and characteristics, pain scales and other valid questionnaires of function, quality of life, mood etc).
Information that may be evidence of a pathology is used to support a ‘working diagnosis(es)’, aka ‘clinical diagnosis(es)’. Adequate evidence can be used to justify surgical intervention for a.) ‘further evaluation and confirmation’ of a working diagnosis and/or b.) a patient with high probability to benefit from the procedure.
What’s got my knickers in a twist today?
Phenomenal advances in medicine have improved our understanding of diseases, imaging technology, surgical instruments and optics. Change is occurring parallel to shrinking incision sites (minimal invasive). Unfortunately, pockets of medicine, as applied to specific diseases, are not progressing well. In fact, for some lesser known diseases, the rapid rate of progress with minimally invasive procedures is a double-edged sword.
Endometriosis is not one of these lesser known diseases. It’s a common disease, affecting an estimate 1:10 cis-female across the globe.6 But, awareness and knowledge remains poor and heavily debated. Persistent myths, misinformation and outdated guidelines still contribute to delayed diagnosis. A 2015 questionnaire completed by 4,358 residents of the USA with endometriosis reported an average of 11.3 yrs from first symptoms to diagnosis.7
Knowing this, consider the additional delay for those who do not present with the more common symptoms associated with endometriosis: heavy, painful periods and painful sex. Also consider those who have been diagnosed with endometriosis but have symptoms remote to their ‘lady parts’. In most cases, their care team, and often the individual, don’t associate symptoms with endometriosis that often results in misdiagnosis or dismissal.
So, what does endometriosis have to do with lesser-known diseases and a double-edged sword that comes with progress? In most instances, progress is good, right? Smaller incisions, better surgical vision and tools leads to less intra-operative trauma, shorter hospital stays and less pain during recovery, right? Yes. These are ALL perks of progress. However, I want to talk about a smaller portion of persons affected by endometriosis yet is virtually unknown or, in some minds in medicine, doesn’t exist: Thoracic Endometriosis (TE).
Thoracic Endometriosis (TE) and its manifestations are still considered rare. 8,9 (refer to Video Animation Lesson 2: Is Extrapelvic Endometriosis Rare). Very little, if any, education about the disease is included in curricula (medical school, allied healthcare professional programs and post-professional continuing education). The advent of Video-assisted thoracoscopy (VATS), has expanded the population eligible for surgical investigation that would otherwise be denied or face an extensive post-operative recovery of a thoracotomy. VATS has also prompted earlier investigation among specific patient populations. Here lies the double-edged sword: As populations whom are candidates for VATS expand, persons with TE whom are misdiagnosed with another condition (most oft PSPT) can lead to a care plan that does more harm than good. The lack of awareness, or acknowledgement of the disease, its pathogenesis, presentations, limitations of imaging, treatment and histological staining techniques used to identify the disease are barriers to best outcomes. Another, larger portion of the TE population are comprised of persons who do not develop lung collapse(s), bloody fluid in the chest cavity or cough up blood, yet experience cyclic symptoms, that can be incapacitating. Based upon the current criteria outside of a few multidisciplinary endometriosis surgical centers, this population will never receive a VATS.
At present, criteria for VATS contributes to delay in diagnosis of TE. Adjustment in the criteria for VATS among this population will reduce the lag to diagnosis. But, if concurrent curricula and surgical training do not coincide these changes, continued misdiagnosis and improper interventions can result in greater complications with missed or incomplete removed disease. The double-edged sword.
Although not a debate, I will use this blog as my soapbox. I campaign for an adjustment of criteria for use of Video-Assisted Thoracoscopy (VATS), when suspicion is high for the presence of Thoracic Endometriosis. But, I specify here and now, this proposal is limited to persons with documented endometriosis in the abdominopelvic cavity and symptoms highly suspicious for TE. The proposal includes development and validation of a screening questionnaire that establishes probabilities of disease, and a scoring system or other objective scale to meet criteria for VATS among persons without a history of manifestations associated with TES. The proposal extends to include mandatory education of numerous first-contact healthcare professionals. These details are explained below, in addition to facts about the disease and history of attempts to establish probabilities for presence of TE for earlier diagnosis.
It is important to clarify: A few multidisciplinary endometriosis excision surgical centers have different criteria for VATS. Based upon experience and high volume persons with suspicion for TE, a prior manifestation may not be necessary. This differs from ‘the rest of the world’.
For ‘the rest of the world’, non-gyn providers and non-endometriosis focused gyn, there is little awareness about endometriosis of the respiratory system. Lack of awareness and ability to recognize direct and indirect ‘evidence’ delays, and often prevents attainment of gold standard procedure, to assess and treat disease in the chest cavity. Although a portion of persons with thoracic endometriosis (TE) are identified and undergo a VATS, it is only a small portion of persons with the disease. A lack of surgeon knowledge of disease’s presentations, theories of pathogenesis and histological testing can lead to missed disease and, if there is a history of manifestations (ie lung collapse), preventive measures are often applied that increases surgical complexity if a further surgery is necessary.
I mentioned ‘small portion’ immediately above. I’m referring to the fact that only a small portion of persons with TE will experience a manifestation, or have accessed a multidisciplinary endometriosis surgical excision center. Some with TE experience manifestations early in their journey, others develop them later in their journeys after onset of symptoms. But, the majority of persons with TE never experience a lung collapse, blood in the chest cavity or coughing of blood yet endure cyclic, to daily presence of symptoms that can be incapacitating.
Knowing this, let’s take a look at the current guidelines and criteria for VATS:
Objective evidence is a significant factor. Treatment for PSPT is one of the most common reasons for VATS: Further, SPT is THE most common manifestation of TE.1 The general standard for VATS include: 10

We Know: The manifestations of TES (excludes CCP in the expanded description) account for a smaller portion of the population with TE. Hence, a larger portion of persons with TE do not experience a manifestation or may be latent. These are persons with CCP.
Now, we KNOW the risk of recurrent SPT’s due to endometriosis (CP or ERP is very high).11 We also know that up to 1 in every 3 SPT’s among cis-females have been identified as CP or ERP.2,4 Given the statistics, the first recommendation would be to assess for evidence (Objective, Observational and Subjective-Objective) upon the FIRST episode that raises suspicion that endometriosis is highly probable.

A recent review of VATS for investigation of CP only included subjects with a history of at least 2 SPT’s of same lung. It is unclear if this standard was due to a lack of published cases of subjects with a single CP or if persons were only diagnosed following a recurrent SPT and/or a single SPT was not sufficient evidence to justify a VATS.12
The attempt to lower the threshold of evidence to justify VATS for persons with CP/ERP, and others with TE, is slowly publishing data to support it.
Data which identified specific characteristics among persons with (1+) CP/ERP established probabilities for the presence of TE and justify VATS.2,13
“Diaphragmatic MRI could also aid in the management of women presenting with a first catamenial pneumothorax. Thoracic surgery is usually only performed in the case of recurrent catamenial pneumothorax, although some authors have suggested performing surgery after the first episode of catamenial pneumothorax when it is associated with high clinical suspicion of endometriosis. In such a context, a positive diaphragmatic MRI, especially if focal liver herniations are observed, would strongly reinforce the indication to initiate specific surgical management. (Rousset P et al. 2016)
Characteristics independent of any history of pneumothorax that were highly predictive for thoracic endometriosis included:

A few years later, another attempt to improve diagnosis of thoracic endometriosis based upon occurrence of one or more SPT and patient traits. A score of 12 or greater (of a maximum 18 points) was 93.5% sensitive and 89.4% specific for endometriosis.4

“Patients with diagnosed pelvic endometriosis showed a significantly higher rate of endometrial implants and histologically confirmed endometriosis lesions (in the chest cavity, through VATS) than patients without pelvic endometriosis.” (Bricelj K et al, 2017)
“The main intraoperative findings were endometrial implants, diaphragmatic perforations, or blebs/bullae. Compared with patients without pelvic endometriosis, patients with pelvic endometriosis had a significantly higher rate of findings of endometrial implants. There was a significant difference in the finding of histologically confirmed thoracic endometriosis between patients with pelvic endometriosis and patients without. Regarding treatments, 68.1% of patients had hormone treatment, and 62.6% chose combination therapy with VATS and hormone treatment. Overall, 26.9% had a recurrence.” (Azuma Y and Iyoda A, 2017)
As mentioned earlier, the majority of persons with TE do not develop a manifestation. In addition, a spontaneous pneumothorax (catamenial or ERP) is the most common manifestation that occurs. These spontaneous pneumothoraxes are frequently misdiagnoses as Primary Spontaneous Pneumothorax. With introduction of VATS, improvements since inception and data accumulating for outcomes, suggestions have been made to lower the criteria for VATS among those with clinical diagnosis of PSPT:
“Over the years, there have actually, already been a number of clinical series suggesting that surgery after a first episode of PSP may offer advantages over non-surgical management. Nowadays, the answer is further swinging increasingly in favor of surgery because advances in the performance and understanding of surgical pleurodesis have emerged that further improve outcomes for PSP patients.” (Sihoe A DL et al, 2015)
Do you see the concerns I see? If the pendulum IS swinging toward adjusting the criteria to ‘first event’ this will impact persons with 1st CP/ERP. The potential for earlier diagnosis is wonderful BUT, awareness and education about TE, identification, removal and staining for disease and, most importantly, remove ALL disease and repair all defects, diaphragm included is a big concern.
Wait! I am not ready to get off my soapbox yet. My campaign does not end there.
We know, the greater proportion of persons with TE NEVER experience a manifestation. They never meet the current criteria (1 or 2 SPT’s) for VATS. However, evidence is accumulating to support adjusting the criteria for VATS for this population. Some of these studies have already been discussed above. Here, we highlight common complaints reported among persons with TE and include: a history of cyclic scapular and/or chest pain; confirmed endometriosis in the pelvic and/or history of symptoms suggestive of undiagnosed pelvic endometriosis. In attempt to diagnose TE earlier, a few studies have analyzed characteristics among persons with CCP and no history manifestations who’ve undergone VATS. A small study of 20 persons with CCP were referred to a Thoracic Department for consideration of VATS to assess for TE. After screening, 11 persons underwent VATS. Their duration of CCP was 1-6 years. Endometriosis and/ or endometriosis-induced diaphragm defects were identified in 8/11 subjects ((72.7%). Results support the use of VATS when suspicion for TE is high. Unfortunately details of inclusion criteria for surgery were not included in the publication.5
Survey:
We conducted a short survey of women who underwent a VATS for suspicion of TE, and did not have a history of any confirmed manifestations.16 A link to the survey was posted in our education and support group for persons with, or suspect for endometriosis outside of the female reproductive organs and surrounding tissues. Criteria for persons to complete the survey: 1.) History of Pelvic Endometriosis 2.) Symptoms suggestive of TE prior to surgery 3.) NO history of spontaneous pneumothorax, hemothorax or coughing of blood with menses. 4.) Confirmed Diagnosis of TE after VATS procedure.
Responses were collected over 4 days in the month of July. A 10 question, anonymous survey with self-reported experiences to include: duration of symptoms prior to diagnosis, visits to the emergency dept for symptoms associated with TE prior to diagnosis, imaging, surgical intervention in tandem or performed on separate dates and operative findings of disease by location.

Only 4/7 respondents underwent a single surgical event for assessment and treatment for their endometriosis. For these individuals, a VATS was completed during a single surgical event in coordination with a laparoscopy. 3/7 respondents underwent surgical procedures on two separate dates. All 3 underwent a laparoscopy; 2/3 reported endometriosis present on the abdominal side of diaphragm and 1/3 reported no diaphragm disease was identified during laparoscopy. All 3 persons had unresolved symptoms suggestive of TE. These 3 persons underwent a separate VATS at a later date with identification of chest side endometriosis.
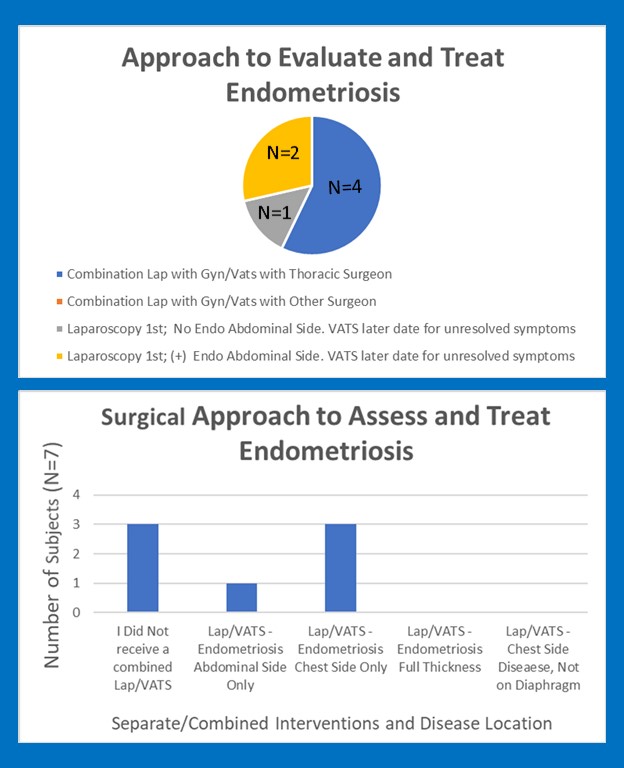
Accumulations of literature has determined that the distribution of endometriosis within the chest cavity, from most to least common areas are the diaphragm > parietal pleura (chest wall and covering of diaphragm) > visceral pleura (outer surface of lung) > lung parenchyma > bronchi airway system.
Based upon location and properties of each imaging technique, MRI is best suited to detect presence of endometriosis. However, MRI is significantly more expensive than CT Scan, doesn’t expose the person to radiation but has been reported to miss 18%-22% of lesions of diaphragm. The majority of our respondents (5/7) received at least (1) CT Scan; only 2/7 received MRI Scan and (2) persons did not receive any imaging of soft tissue structures (neither CT Scan or MRI). Of (7) responders, we divided CT Scan and MRI imaging into groups to determine if the presence of active menstruation and/or symptoms on the day of image appoint were associated with identification of abnormalities on imaging. In our sample survey of (7), there were NO abnormalities identified on any images, regardless of the type of imaging modality. The timing of active menses did not impact ability to identify presence of disease.

Although our sample is limited to the self-reported experience of (7) persons, the survey supports the findings of Bobbio et al (5) discussed above. In their study, 11/20 persons referred to the Thoracic Surgery Department for Catamenial Chest Pain (CCP) underwent VATS.
Findings of disease and/or defects suggestive of TE were found in 8/11 persons. It’s unclear if preoperative images were taken or their results. Similarly, to our survey, no prior history of pneumothorax, hemothorax or coughing blood with menses were reported.
Imaging:
I know what you are thinking. ‘Even though they haven’t had a manifestation, surely the disease would be detected on imaging!’. Nope. Imaging is not 100% sensitive for endometriosis of the respiratory tissues.
“…a negative MR examination does not exclude the diagnosis of diaphragmatic endometriosis in women with suggestive symptoms and would not eliminate the need to surgically investigate the upper abdomen and diaphragm.” (Foti PV et al, 2018)
The inherent limitations of current radiology technology, coupled with limited understanding of TE, experience and knowledge of clinicians to consider the pathology, which tests and when they should be used, and radiologist ability to identify disease vs artifact and, last, consider factors of image timing to menses, lesion shape, size and location, lesion composition coupled with the type of imaging, to understand the probability that disease is present despite an unremarkable image that did not detect a disease process.
“The MRI appearance of endometriotic lesions is variable and depends on the quantity and age of hemorrhage, the amount of endometrial cells, stroma, smooth muscle proliferation and fibrosis.” (Foti PV et al, 2018)
The provider must know probabilities for disease location taking consideration for location and characteristics of the persons symptoms; the type of imaging that best highlights the tissues most speculated, the sensitivities of these imaging techniques to identify endometriosis AND factors that affect sensitivity (lesion composition, shape, size, timing of imaging to menses etc)

MRI allows the diagnosis of diaphragmatic endometriosis implants with a sensitivity of 78–83%.13
“MRI underestimated the number of diaphragmatic lesions and failed to detect small superficial nodules and small diaphragmatic holes. Thus, a negative MRI examination does not exclude the diagnosis of diaphragmatic endometriosis in women with suggestive symptoms.” (Rousset P et al, 2016)
So, here lies the problem: Most with TE don’t develop a manifestation. If they do, its often years to decades later. Imaging is evolving but sensitivity to detect disease but a significant portion with TE have negative imaging. A few high quality studies present evidence that supports lowering evidence threshold to a single manifestation (objective) and presence of specific subjective-objective evidence that justifies a VATS. The subjective-objective evidence extracted from their findings were used to determine if specific subjective-objective evidence was significant to justify VATS among persons without history of any manifestations. The results: Yes!
The use of VATS has been clearly identified as the gold-standard procedure to diagnose and treat endometriosis of the chest cavity. The focus must be on altering the types of evidence based upon associations and probabilities for disease presence for more persons with the disease to experience benefits from the intervention.
“Video-assisted thoracoscopic surgery (VATS) with removal of all ectopic endometrial tissue, closure of diaphragmatic defects, and pleurectomy is considered the reference standard for both definitive diagnosis and treatment. A multidisciplinary approach of combined VATS and laparoscopy is recommended to treat pelvic, diaphragmatic and thoracic endometriosis in a single procedure.” (Chamie LP et al, 2018)
Further, as literature reveals, there are characteristics among persons with symptoms suspicious for TE that have been associated with presence of disease among cis-women with one or more confirmed SPT. These characteristics have been associated among small sample size studies of cis-women with history of CCP without manifestations.
Would these persons benefit from VATS? Do the benefits outweigh the risks?
I was unable to locate literature, including two recent systematic reviews of VATS, which reported complication rates in its use to assess and treat thoracic endometriosis.12,14
Are the financial costs worth a potential improved quality of life?
We have reviewed the current standards of evidence among providers and their facilities whose practices are not part of an endometriosis-focused excision team. These higher norms were compared to the few surgical centers for endometriosis who routinely perform VATS on patients with symptoms highly suggestive of TE, even without a history of manifestations. If the degree of evidence remains at 2 occurrences of an event on the same side OR 1 occurrence plus abnormal image/unresolved air leak or fluid production OR 1 occurrence plus presence of specific characteristics that suggest a high probability of endometriosis, thus justify VATS….then the majority of persons with TE will continue to suffer without gold-standard diagnosis and intervention.
How do we get from here to there? Do you see the double-edged sword?
It is a lot to ask of those who work in multidisciplinary surgical center, excising endometriosis throughout the body on a daily basis, but, development of subjective-objective characteristics, their presence/absence among persons with CCP, and surgical findings may lead to a screening protocol for use among other other healthcare professionals and non-endometriosis to identify a larger portion of persons who will benefit from VATS whom are unlikely to be unidentified or only after development of a latent manifestation. Even then, TE may not be suspect.
Development of a screening protocol also requires awareness and education across the healthcare spectrum, recognition for referral to those in specialty centers. Until recognition of TE signs and symptoms, understanding of the disease process, presentation and limits of imaging for diagnosis are better understand, can the process of surgical skills to address disease in the chest be approached to surgeons outside of endometriosis focused, multidisciplinary surgical excision centers. It is important to acknowledge the risk of inexperienced surgeons performing VATS to assess and treat manifestations or CCP that may miss presence of disease, remain symptomatic. For persons who may have developed a pneumothorax, procedures applied to prevent recurrent episodes increases the complexity of a future attempt to remove missed disease.
…”women presenting thoracic pain during menses should be advised to consult in case of abnormal or unusual thoracic pain and dyspnea. Better knowledge about this disease among specialists in Emergency Medicine, Pneumology, Gynecology and Internal Medicine could help in reducing the delay in diagnosis so that these young women can receive timely treatment…” (Rousset-Jablonski C et al, 2011)
Trish (July 28th, 2020)
References:
Joseph J, Sahn SA. Thoracic endometriosis syndrome: new observations from an analysis of 110 cases. Am J Med. 1996; 100:164-70.
Roussett-Jablonski C, Alifano M, Plu-Bureau G, Camilleri-Broet S, Rousset P, Regnard JF, Gompel Catamenial pneumothorax and endometriosis-related pneumothorax: clinical features and risk factors. Hum Reprod. 2011; Vol 0 (0);1-8. Doi:10.1093/humrep/der189
Fukuoka M, Kurihara M, Haga T, Ebana H, Kataoka H et al. Clinical characteristics of catamenial and non-catamenial thoracic endometriosis-related pneumothorax. Respirology.2015;20:1272-1276. DOI: 10.1111/resp.12610
Haga T, Kataoka H, Ebana H, Otsuji M, Seyama K et al. Thoracic Endometriosis-Related Pneumothorax distinguished from Primary Spontaneous Pneumothorax in Females. Lung.2014. DOI:10.1007/s00408-014-9598-1.
Bobbio A, Canny E, Lupo AM, Lococo F, Legras A et al. Thoracic Endometriosis Syndrome Other Than Pneumothorax: Clinical and Pathological Findings. Am Thorac Surg.2017;104:1865-1871.
World Endometriosis Society. Website: Endometriosis.org (accessed 07/27/2020)
Fourquet J, Sinaii N, Stratton P, Khayel F, Alvarez-Garriga C et al. Characteristics of women with endometriosis from the USA and Puerto Rico. J Endometr Pelvic Pain Disorder.2015;7(4):129-135. DOI: 10.5301/je.5000224
Adesanya OA, Kolawole OE. Thoracic endometriosis syndrome: Cutting the gordian knot – A case report and review of the literature. Int’l J Surg Case Rep. 2020;66:68-71.
Alifano M. Catamenial Pneumothorax. Rare Disease Database. National Organization of Rare Disorders. NORD. Website: raredisease.org. Accessed 07/27/2020.
Paliouras D, Barbetakis N, Lazaridis G, Baka S, Mpoukovinas I et al. Video-assisted thoracic surgery and pneumothorax. Review Article. J Thorac Dis.2015;7IS1):S56-S61. DOI: 10.3978/j.issn.2072-1439.2015.01.35
Mehta C, Stanifer BP, Fore-Kosterski S, Odell DD, DeCamp MM, Bharat A. Primary spontaneous pneumothorax in menstruating women has high recurrence. Ann Thor Surg.2016. DOI: 10.1016/j.athoracsur.2016.04.069.
Bricelj K, Srpcic M, Razem A, Snoj Z. Catamenial pneumothorax since introduction of video-assisted thoracoscopic surgery. A systematic review. Wein Klin Wochenschr. 2017. Doi: 10.1007/s00508-017-1237-4.
Rousset P,ory J, Rousset-Jablonski C, Hugon-Rodin J, Regnard JF et al. MR Diagnosis of diaphragmatic endometriosis. Eur Radiol.2016. DOI: 10.1007/s003300-016-4226-5
Azuma Y, Iyoda A. Results of treatment for catamenial pneumothorax since the introduction of video-assisted thoracoscopic surgery: a systematic review. Video Assist Thorac Surg.2017;2:71
Sihoe A DL, Yu P SY, Yeung J WL. Primary pneumothorax: Should surgery be offered after the first episode? World J Respirol.2015;5(1):47-57.
Internal Survey. Members of Extrapelvic Not Rare Endometriosis Education and Support Group. Data collection July 20-27, 2020.
Foti PV, Farina R, Palmucci S, Vizzini IAA, Libertini N et al. Endometriosis: clinical features, MR imaging findings and pathological correlation. Insights into Imaging.2018;9:149-172.
Chamie LP, Ribeiro DMFR, Tiferes DA, de Macedo Neto AC, Sarafinia PC. Atypical Sites of Deeply Infiltrating Endometriosis: Clinical Characteristics and Imaging Findings. Radiographics.2018;38:309-328.
